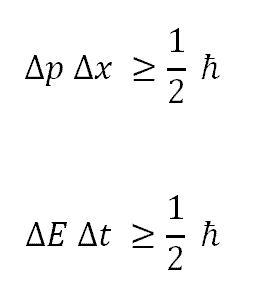Trích:
|
Nguyên văn bởi TNT_TNT
cho đệ hỏi trong nglý bất định Heisenberg thì chính xác là denta x nhân denta px >= h hay là h/2pi (tức là h ngang)
|
The uncertainty relations can be written a little more precisely in the shorthand of mathematical symbols. But first we must define what these symbols stand for. Here are their definitions:
Dq is the uncertainty or imprecision (standard deviation) of the position measurement.
Dp is the uncertainty of the momentum measurement in the q direction at the same time as the q measurement.
DE is the uncertainty in the energy measurement.
Dt is the uncertainty in the time measurement at the same time as the energy is measured.
h is a constant from quantum theory known as Planck's constant, a very tiny number.
p is pi from the geometry of circles.
> means "greater than or equal to"
Putting these symbols together, the two uncertainty relations look like this:
Dp Dq > h / 4p
DE Dt > h / 4p
Let's say you measure the position of a moving electron with such great accuracy that Dq is very small. What happens to the precision of the momentum Dp, which you measure at the same instant? From the first relation, we have
Dp > h / 4 p Dq
http://www.aip.org/history/heisenberg/p08a.htm